Mysterious Sunflower Syndrome is Spotlighted at Massachusetts General Hospital
July 24, 2019
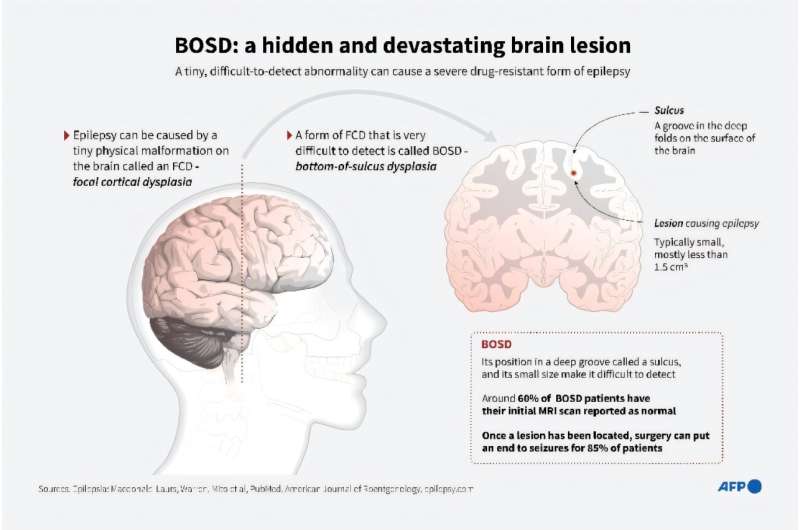
Sunflower syndrome [is] a rare form of epilepsy that, in a new study, researchers at Massachusetts General Hospital are working to understand – and treat. Sunflower syndrome compels those who suffer from it to turn toward bright light and wave one hand, fingers spread, in front of their eyes. It’s usually the same hand every time. This unique behavior is coupled with abrupt lapses in consciousness that last several seconds.
“About half of the kids do it both indoors and outdoors. It’s pretty crippling,” said Dr. Elizabeth Thiele, director of the pediatric epilepsy program at MassGeneral Hospital for Children. She has fielded calls from patients around the world and seen dozens of them, including a 7-year-old boy who obsessively drew pictures of the sun.
She said her latest work measuring brain activity in sunflower patients, which has not been published, shows only a fraction of a second elapses between the start of the hand-waving and the onset of the seizure, not nearly long enough for the action to create a seizure. It demonstrates, she said, that the waving is part of the seizure and is not provoking it.
Thiele is launching a four-month study of 10 patients to test whether an appetite suppressant banned two decades ago, Fenfluramine, might be effective in controlling the syndrome. Fenfluramine, often combined with another medication and sold as a popular diet pill known as Fen-Phen, was pulled from sales after reports the drugs caused significant heart damage.
But Fenfluramine was not banned in Belgium, and researchers have demonstrated that low doses taken for years have significantly reduced seizures in another drug-resistant form of epilepsy, Dravet syndrome, without producing heart damage. Now, federal regulators are considering whether to allow Fenfluramine back on the market to treat Dravet in the United States, possibly preparing the way for sunflower patients.