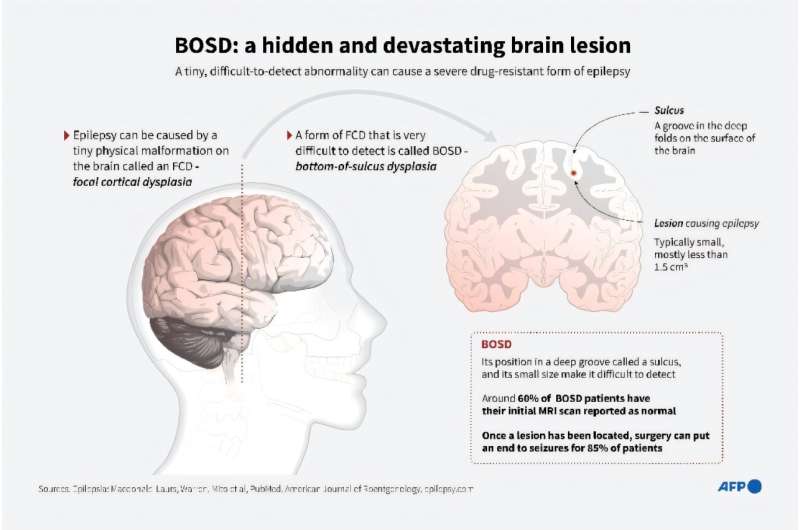
Ovid Therapeutics Announces Initiation of Two Phase 2 Clinical Trials of OV935/TAK-935 for Pediatric Patients with Rare Epilepsies
September 24, 2018
Ovid Therapeutics Inc. announced initiations of the Phase 2 ELEKTRA and ARCADE trials for OV935/TAK-935 in pediatric patients with rare epilepsies. The company reports that the first patient has been randomized to receive either OV935 or placebo in the ELEKTRA trial, and that patient screening is underway for the ARCADE trial.