Stoke Therapeutics Observational Study of Children and Adolescents Living with Dravet Syndrome
August 20, 2019
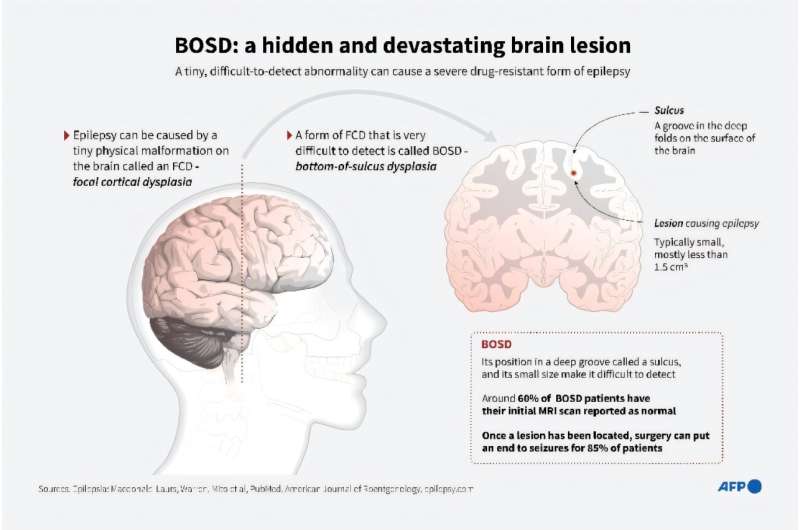
Stoke Therapeutics, Inc. announced enrollment of the first patient in an observational study of children and adolescents ages 2 to 18 with Dravet syndrome. BUTTERFLY is a two-year observational study that is designed to evaluate seizure frequency and non-seizure comorbidities associated with the disease, including motor and speech impairment, intellectual and developmental disabilities, behavioral deficits and abnormal sleep patterns. Data from the study will support clinical development plans for the company’s lead therapeutic candidate, STK-001, an investigational new treatment for Dravet syndrome.