Multicenter Clinical Study of Long-Term Brivaracetam Use Finds Brivaracetam Effective and Well-Tolerated in Patients with Drug-Resistant Epilepsy
April 5, 2019
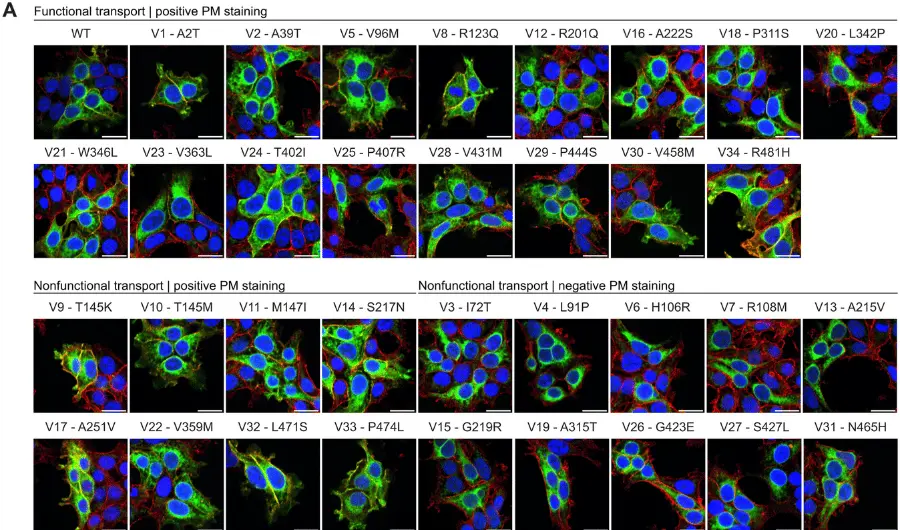
Researchers assessed long-term effectiveness and tolerability of brivaracetam in patients with focal epilepsy. According to this multicenter retrospective study, brivaracetam was effective and well-tolerated in a large population of patients with predominantly drug-resistant epilepsy. There were no unexpected adverse events (AEs) over 1 year. The most frequently reported AEs were somnolence, irritability and dizziness.