Zogenix Submits New Drug Application to FDA and Marketing Authorization Application to European Medicines Agency for FINTEPLA® for the Treatment of Dravet Syndrome
February 11, 2019
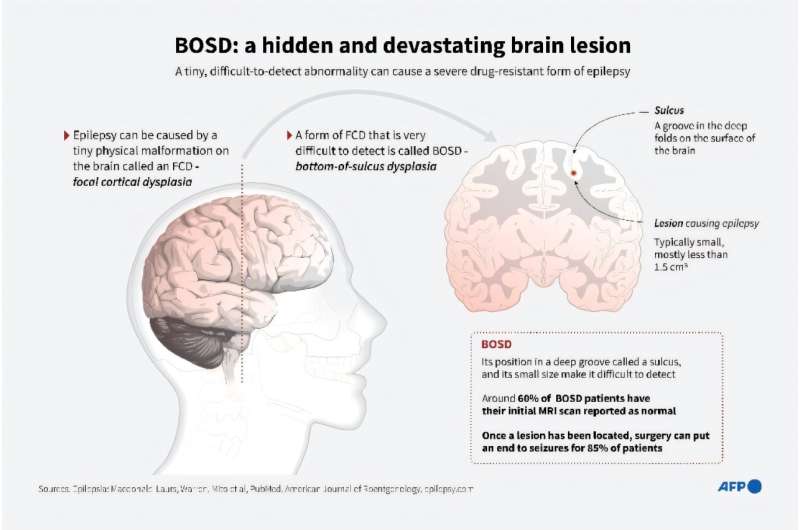
Zogenix, Inc. announced it has completed its rolling submission of a New Drug Application (NDA) to the FDA and submitted a Marketing Authorization Application (MAA) to the European Medicines Agency (EMA) for FINTEPLA (ZX008, low-dose fenfluramine) for the treatment of seizures associated with Dravet syndrome. Dravet syndrome is an intractable and difficult-to-treat epilepsy that begins in infancy and is associated with frequent, severe, and potentially life-threatening seizures, developmental delay, and cognitive impairment.
Both applications are based on data from two pivotal Phase 3 trials in Dravet syndrome and an interim analysis from an ongoing open-label extension study, which included 232 patients treated for up to 21 months.