Understanding Epilepsy Main Menu
Table of Contents:
- What are Infantile Spasms
- Symptoms and Signs of Infantile Spasms
- Infantile Spasms and Seizures in Infants
- What causes Infantile Spasms?
- Who can suffer from Infantile Spasms?
- How Infantile Spasms are Diagnosed
- Tools and Tests for Diagnosis
- What is the long-term prognosis for infantile spasms?
- Treatment Options for Infantile Spasms
- Infantile Spasms FAQ
What are Infantile Spasms
Infantile spasms, also known as West syndrome, are a rare type of seizure disorder that occurs in infants, typically between the ages of 3 to 12 months. These spasms often manifest as sudden, jerking movements of the arms, legs, or trunk, resembling a startle reflex.
Infantile spasms are characterized by subtle infant seizures, which can have significant neurological effects and an atypical EEG pattern; these symptoms can lead to considerable developmental delays and cognitive and physical deterioration. Because these spasms do not look like traditional seizures, primary care physicians and pediatricians can find it challenging to diagnose Infantile spasms.
This often means that effective treatment for affected children is delayed, which can have dire consequences. And for those that are diagnosed accurately, many do not respond to treatment or have adverse side effects.
Signs and Symptoms of Infantile Spasms
The symptoms of infantile spasms may include:
- Sudden jerking movements of the limbs
- Brief moments of stiffness or rigidity
- Loss of developmental milestones
- Regression in cognitive or motor skills
Infantile Spasms and Seizures in Infants
Perhaps the most important way to improve infantile spasms outcomes is to recognize the signs and symptoms and get children suspected of having infantile spasms evaluated by a trained medical provider. Consider using the STOP Infantile Spasms mnemonic developed by ISAN as an important starting point:
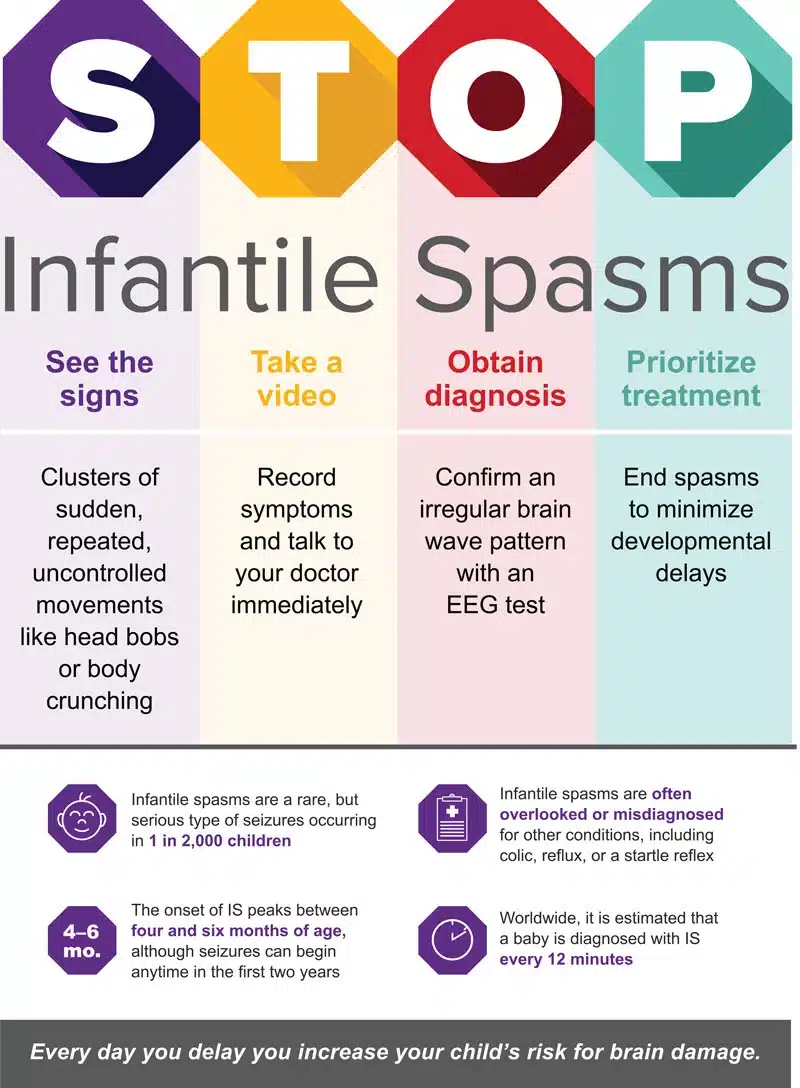
What causes Infantile Spasms?
It is estimated that 2 out of 3 infants with Infantile Spasms have a known cause for their seizures. However, the exact cause of infantile spasms is not always clear. Some causes might include:
- Structural changes in the brain, which may stem from previous injuries like brain infections or oxygen deprivation. Additionally, alterations in brain development, such as cortical malformations or dysplasia, can also contribute to their occurrence.
- Genetic factors may play a role in causing infantile spasms, with several genes being linked to this condition.
- Metabolic issues can disrupt brain function and trigger spasms in some cases.
- Some infants experience spasms without any apparent injury and have been progressing typically in their development.
- There is no indication that factors such as family history, the baby’s gender, or immunizations are connected to the occurrence of infantile spasms.
Who can suffer from Infantile Spasms?
Infantile spasms primarily affect infants, typically between the ages of 3 to 12 months. However, it’s important to note that while they are most common in infants, older children and even adults can also experience spasms in rare cases. Nonetheless, the term “infantile spasms” specifically refers to the occurrence of these seizures in infants.
How Infantile Spasms are Diagnosed
Diagnosing infantile spasms often involves a comprehensive medical evaluation, including a review of the infant’s medical history, a physical examination, and neurological tests. Healthcare providers may also use imaging studies, such as MRI or CT scans, to assess for any underlying brain abnormalities.
Tools and Tests for Diagnosis
Electroencephalography (EEG) is a key tool in diagnosing infantile spasms. This test measures the electrical activity in the brain and can help identify abnormal patterns associated with seizures. Video EEG monitoring may also be used to capture and analyze the infant’s behavior during a seizure.
What is the long-term prognosis for infantile spasms?
Children affected by infantile spasms face the potential of developing additional seizure types as they grow older. They may also be susceptible to conditions like autism and cognitive delays. However, early detection and treatment can aid in mitigating these risks.
Treatment Options for Infantile Spasms
Medications
The primary goal of treatment for infantile spasms is to control seizures and minimize their impact on the infant’s development. Antiepileptic medications, such as adrenocorticotropic hormone (ACTH) or oral steroids, are commonly used to suppress seizure activity and improve outcomes.
Therapies
In addition to medication, infants with spasms may benefit from early intervention services, such as physical therapy, occupational therapy, and speech therapy. These therapies can help address developmental delays and support the infant’s overall growth and development.
Surgical Interventions
In some cases, surgery may be considered as a treatment option for infantile spasms, particularly if there is an underlying structural abnormality in the brain that can be corrected. Surgical procedures may involve removing or resecting the affected area of the brain to reduce seizure activity.
Infantile Spasms FAQ
-
What do infantile spasms look like?
Infantile spasms can be subtle or obvious. In the subtlest cases, these seizures may take the form of a slight drop of the head or other type of involuntary movement. They might also look like the classic Moro reflex, in which an infant reaches both arms out at the same time. Other infantile spasms may involve the infant extending or jerking its arms, legs, and neck at the same time. These spasms can occur as a single event, or may occur in clusters of several spasms. They often occur in the morning or after the child wakes from a nap.
To view videos of children with infantile spasms, please visit the Infantile Spasms Action Network.
-
Are infantile spasms genetic?
There are some genetic links to infantile spasms. Mutations, deletions, or abnormalities in certain genes have been associated with infantile spasms. These gene abnormalities associated with the presentation of infantile spasms can lead to many different possible changes in the brain depending upon the gene involved – changes such as brain malformations, errors in metabolism, changes in brain excitability, or other genetic disorders (such as tuberous sclerosis). (Paciorkowski et al., 2011)
Importantly, there is a lot left to learn about the underlying mechanisms of infantile spasms. Therefore, continued research into the underlying causes of infantile spasms is essential.
-
Where can I get more information on infantile spasms?
Child Neurology Foundation: An organization that provides support and education for caregivers and children with neurologic conditions, including the promotion of infantile spasms awareness and providing information on the Infantile Spasms Action Network.
Tuberous Sclerosis Alliance: An organization committed to supporting tuberous sclerosis research and providing education and support for individuals and families affected by the disorder.
Stay in touch
Sign up for our email list and get the latest epilepsy news right to your inbox.







