Re-Annotation of 191 Developmental and Epileptic Encephalopathy-Associated Genes Unmasks De Novo Variants in SCN1A
December 5, 2019
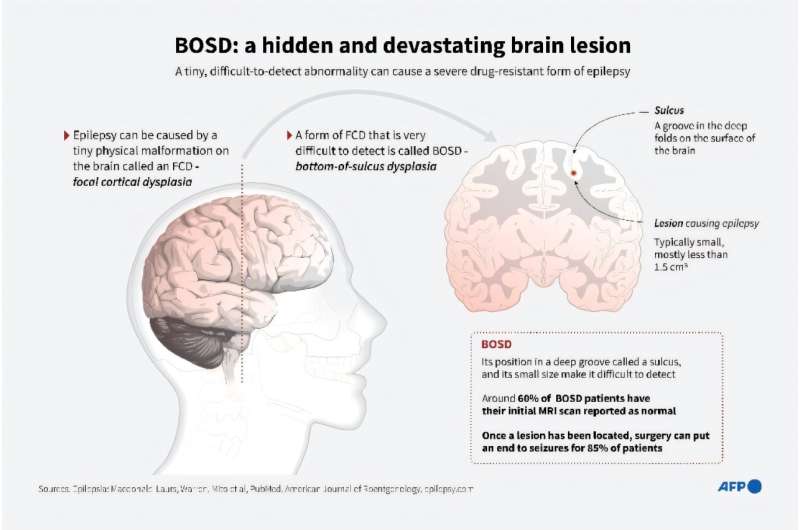
The developmental and epileptic encephalopathies (DEE) are a group of rare, severe neurodevelopmental disorders, where even the most thorough sequencing studies leave 60–65% of patients without a molecular diagnosis. Here, researchers explore the incompleteness of transcript models used for exome and genome analysis as one potential explanation for a lack of current diagnoses. Therefore, we have updated the GENCODE gene annotation for 191 epilepsy-associated genes, using human brain-derived transcriptomic libraries and other data to build 3,550 putative transcript models. These annotations increase the transcriptional “footprint” of these genes by over 674 kb.
Using SCN1A as a case study, due to its close phenotype/genotype correlation with Dravet syndrome, the team screened 122 people with Dravet syndrome or a similar phenotype with a panel of exon sequences representing eight established genes and identified two de novo SCN1A variants that now – through improved gene annotation – are ascribed to residing among our exons. These two (from 122 screened people, 1.6%) molecular diagnoses carry significant clinical implications.
Furthermore, the team identified a previously classified SCN1A intronic Dravet syndrome-associated variant that now lies within a deeply conserved exon. These findings suggest the potential gains of thorough gene annotation in improving diagnostic yields for genetic disorders.