Leadership in SUDEP
The NIH now funds nearly $20M annually in SUDEP research. Leaders at the NIH credit CURE Epilepsy’s early, modest projects as the initial spark that generated exponential interest and investment at the federal level. That is the type of foundational impact that CURE Epilepsy has year over year.
CURE Epilepsy is unwavering in our commitment to end SUDEP.
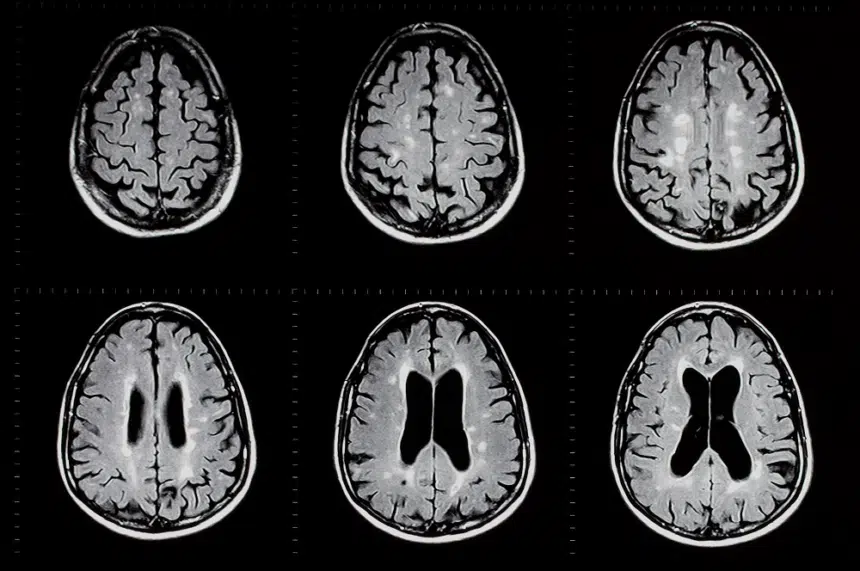
SUDEP is one of the most devastating but least talked about consequences of epilepsy. Every year it impacts over 1 in 1,000 people living with epilepsy (Sveinsson et al. 2020 Neurology), and countless more when you consider these individuals’ families and friends who suffer untold grief and sadness from the loss of a loved one.
In response to this unacceptable loss, CURE Epilepsy, with the assistance of many parents who were seeking answers, launched the first-ever private research program dedicated to advance the understanding SUDEP and its prevention in the US.
Established in 2004, our cutting-edge SUDEP program impacts include:
- Elevating physician and patient awareness of SUDEP
- Funding research that helped identify a SUDEP biomarker and the development of a seizure alarm that could potentially prevent SUDEP
- Helping to establish respiratory arrest as a leading cause of SUDEP
- Revealing the link between SUDEP and genes found in both the brain and the heart
- Funding four SUDEP registries and multiple animal models of SUDEP in both the US and Canada
- Helping establish generalized tonic-clonic seizures as a clear risk factor for SUDEP
- Supporting the passing of SUDEP reporting legislation in several states
“CURE Epilepsy’s contribution to understanding the risk of sudden death in epilepsy has been early, sustained, and transformative. From a handful of case reports 20 years ago to over 200 scientific publications in 2021, CURE Epilepsy’s support of individual investigators provided the engine to develop advanced basic and clinical collaborators. We all owe CURE Epilepsy our gratitude for its continuing leadership in accelerating this effort.”
Jeff Noebels, MD, PhD Baylor College of Medicine
Current SUDEP Work
SUDEP Risk Assessment and Prevention Initiative
CURE Epilepsy’s latest work in SUDEP is translating scientific insights on biological factors into meaningful clinical progress. Our team science work integrates multi-disciplinary experts across laboratories working on global datasets with an end goal of integrating the discussion of SUDEP into the clinical visit workflow in a way that empowers both patients and doctors to take steps to mitigate risk. With continued investment in research, we can help people understand their relative risk and improve outcomes for people with epilepsy (i.e. prevent SUDEP).
The SUDEP Risk Assessment and Prevention Initiative started in 2024 with the formation of a steering committee of researchers committed to reducing the risk of SUDEP. The initiative is comprised of three components:
The Initiative was kicked off with a gift from the Epilepsy Foundation of America stemming from their Biomarker Challenge. The Cameron Boyce Foundation sponsored the workshop, and other private donors have made our progress possible thus far.
More on this initiative
-
Workshop
- 20 multidisciplinary researchers from around the country and the world came together in New York with an additional 5 attending parts of the 2-day meeting virtually.
- Experts in neurology, cardiology, pediatric sudden death, pulmonology, radiology, biostatistics and more presented their respective information related to SUDEP.
- The group discussed the available data and the best approach to identifying risks and communicating to patients.
Post-workshop, the initiative’s steering committee identified two projects that should be pursued:
- CURE Epilepsy-supported team science research grant mechanism
- Development of a SUDEP risk assessment tool

-
Team Science Project
The SUDEP Clinical Biomarker Team Science Award brings three teams of scientists together to work on advancing the knowledge of risk factors for SUDEP.
- Goal: Translate scientific insight into meaningful progress—improving safety, informing prevention strategies, and offering hope to families living with epilepsy today and in the future.
- Datasets: Teams participating in this project will use existing clinical data to identify the biological factors that place some individuals at higher risk.
- Collaboration: Teams will work together in a highly collaborative model, sharing data, methods, and findings in real time.
-
Risk Assessment Tool
The end goal of the initiative is to develop a tool that will:
- Help healthcare providers have open, informed conversations about SUDEP risk with people with epilepsy and their caregivers in the clinic
- Give a clear, easy-to-understand picture of an individual’s overall SUDEP risk based on known medical and environmental factors
Prompt meaningful conversations about steps that can be taken to reduce risk where change is possible

Data Standardization Project Publications
During the 2020 SUDEP Coalition Summit, which brought together key stakeholders to provide a roadmap for action towards the ultimate goal of ending SUDEP, the need for tools and methods to better characterize and understand the clinical relevance of SUDEP laboratory models was identified. In 2022, the SUDEP Data Standardization Project was initiated to fill this need, providing a standardized data collection and reporting language to help preclinical researchers characterize and understand SUDEP animal models, with the aim of improving translation of laboratory models to the clinical setting. This effort was generously supported by the BAND Foundation.
Harmonizing Epilepsy Research: Common Data Elements